Bharat Biotech and SII Pledge for a Smooth Rollout of Coronavirus Vaccine
January 05, 2021 16:25
(Image source from: Economictimes.indiatimes.com)
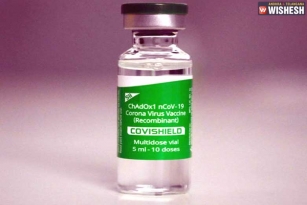
Bharat Biotech and SII Pledge for a Smooth Rollout of Coronavirus Vaccine:- The Indian Drug Regulator already approved two Indian vaccines for emergency rollover but there are a lot of controversies surrounded and speculations going on. Serum Institute of India along with Bharat Biotech pledged to have a smooth rollout of the coronavirus vaccine for the country along with other countries. Mr Adar Poonawala and Dr Krishna Ella jointly on behalf of Serum Institute of India and Bharat Biotech communicated about their combined intent to manufacture a vaccine for the coronavirus. They said that the most important task before them is to save the lives and livelihoods of lakhs of people.
Our pledge towards a smooth roll out of #COVID-19 vaccines to India and the World, along with @SerumInstIndia @adarpoonawalla @SuchitraElla #BharatBiotech #COVAXIN pic.twitter.com/VYbDTkG3NL
— BharatBiotech (@BharatBiotech) January 5, 2021
The two coronavirus vaccines received Emergency Use Authorization (EUA) in the country and they will focus on the manufacturing, supply, distribution. They said that the entire population should get a vaccine of high quality which is safe and efficient. They said that both our companies are fully engaged in this activity and it is the duty of the nation and the world to ensure a smooth rollout of the vaccination. Each of the companies will continue their development activities of the coronavirus vaccine as per the plan. Bharat Biotech and Serum Institute of India said that they are fully aware of the importance of the vaccines for the people of India.